
Ten weeks
after conception, the baby's heart beat can be heard.
Power Point Presentation (broadband)
April 7th: Abortion Trial Transcripts Reveal Truth -
Read the Abortionists' Testimony
(WARNING: GRAPHIC MATERIAL)
It is witchcraft
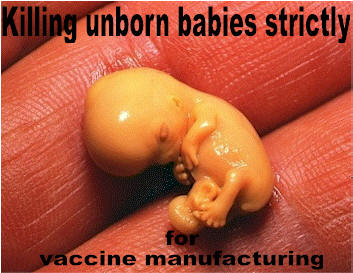 The Chickenpox,
Hepatitis-A and MMR vaccines were developed using aborted fetal cell
lines, MRC-5 and WI-38. This has never been hidden from the
public. When parents take their children to the doctor for inoculations,
who asks for the package inserts? Who asks for a list of the vaccine
ingredients?
Most parents
want to know the risks and possible side effects. Parents are mostly
concerned with the health of their children as some doctors are.
Doctors have been administering the vaccines for years. Yet, how many
of them have ever checked into the ingredients? This has always been
right at their fingertips. What would happen if they did? They would
read that the vaccine contains "MRC-5", "WI-38" (or both) "human diploid
cell lines".
The Chickenpox,
Hepatitis-A and MMR vaccines were developed using aborted fetal cell
lines, MRC-5 and WI-38. This has never been hidden from the
public. When parents take their children to the doctor for inoculations,
who asks for the package inserts? Who asks for a list of the vaccine
ingredients?
Most parents
want to know the risks and possible side effects. Parents are mostly
concerned with the health of their children as some doctors are.
Doctors have been administering the vaccines for years. Yet, how many
of them have ever checked into the ingredients? This has always been
right at their fingertips. What would happen if they did? They would
read that the vaccine contains "MRC-5", "WI-38" (or both) "human diploid
cell lines".
Most parents are not
aware that the particular abortions which contribute the fetal tissue
parts for vaccine manufacturing, were actually intended for vaccine
manufacturing.
 One
if the most highly misunderstood notions among moral theologians and
ethicists is that the abortions involved were not done with the
intention of creating vaccines. In fact, in response to President
Bush’s decision on federal funding for embryonic stem cell research (ESCR),
the USCCB highlights this point as follows:
One
if the most highly misunderstood notions among moral theologians and
ethicists is that the abortions involved were not done with the
intention of creating vaccines. In fact, in response to President
Bush’s decision on federal funding for embryonic stem cell research (ESCR),
the USCCB highlights this point as follows:
“In the present case, human lives were taken in order to provide
cells for research and, in some cases, precisely to qualify for federal
grants; in the case of vaccines, tissues were taken following abortions
performed for unrelated reasons.” -(The
NCCB Secretariat for Pro-Life Activities, Vol. 12, No. 4 Aug-Sept 2001,
The Human Embryo as Research Commodity Special Edition)
It was
the intent of the abortionist and the researcher to destroy these babies
specifically for vaccine manufacturing. But first, the abortions had to
be pre-arranged so the researchers were available to immediately
preserve the tissues.
"In order to sustain 96% of the cells, the live tissue would need to be
preserved within 5 minutes of the abortion"-Dr.
C. Ward Kischer
..."Within one the cells would continue to deteriorate, rendering the
specimens useless."
-[Dr. C. Ward Kischer,
Embryologist and Emeritus Professor of Anatomy; Specialist in Human
Embryology,
University of Arizona College of Medicine (Tucson, Arizona) Personal
interview 7-02, ALL Conference]
1961 Leonard
Hayflick, employed by the Wistar Institute, research facility of
University of Pennsylvania, started working with aborted fetal cell
lines (WI-I through WI-25). Cell strains were derived from the lung,
skin, muscle, kidney, heart, thyroid, thymus and liver of 19 separate
abortions. The entire research was was for the development of viral
vaccine cultivation.
"The
isolation of characterization of human diploid cell strains from fetal
tissue make this type of cell available as a substrate for the
production of live virus vaccines. Other than the economic advantages,
such strains in contrast to heteropoloid cells lines exhibit those
characteristics
usually reserved for normal or primary cells and therefore make the
consideration of their use in the production of human virus vaccines a
distinct possibility"-(L. Hayflick and P.S.Moorhead,
The
Serial Cultivation of Human Diploid Cell Strains, Experimental Cell
Research, 1961, 25, pg 618)
1964 Hayflick
isolates and gives name to the WI-38 cell line derived from the lungs of
a baby girl 3 months gestation.
"This
fetus was chosen by Dr. Sven Gard, specifically for this purpose. Both
parents are known, and unfortunately, for the story, they are married to
each other, still alive and well, and living in Stockholm, presumably.
The abortion was done because they felt they had too many children.
There were no familial diseases in the history of either parents, and no
history of cancer specifically in the families."
-G. Sven, S. Plotkin, K.
McCarthy, Gamma Globulin Prophylaxis; Inactivated Rubella Virus;
Production and Biological Control of Live Attenuated Rubella Virus
Vaccines; Amer J Dis Child Vol 118 Aug 1969
"One of
my duties as a young student in the laboratory in, Stockholm was to
dissect human fetuses from legal abortions and send organs to the Wistar
Institute. Such material was the source of many important studies of
cell lines of the Institute, such as Leonard Hayflick's study of WI-38"
-Norrby,
Erling "Listen to the Music: The Life of Hilary Koprowski (review)",
Perspectives in Biology and Medicine - Volume 44, Number 2, Spring 2001,
pp. 304-306
Preying on the fear that 20%-25% of pregnant
women infected with Rubella pass it to their unborn children, possibly
causing defects, doctors encouraged pregnant women to have their unborn
children aborted during the 1964 epidemic. The first 26 aborted
babies were unaffected. The live rubella virus was found in the 27th
killed unborn baby.
"Explant cultures were made of the
dissected organs of a particular fetus aborted because of rubella, the
27th in our series of fetuses aborted. This fetus was from a
25-year-old mother exposed to rubella 8 days after her last menstrual
period. 16 days later she developed rubella. The fetus was surgically
aborted 17 days after maternal illness and dissected immediately.
Explants from several organs were cultured and successful cell growth
was achieved from lung, skin, and kidney. It was then grown on WI-38,.
The new vaccine was tested on orphans in Philadelphia"
-American
Journal Diseases of Children; Virus Production and Biological Control
of Live Attenuated Rubella Virus Vaccines, Vol. 118 Aug 1969;
Attenuation Of RA273 Rubella Virus; Studies of Immunization With Living
Rubella Virus; Arch J Dis Child vol 110 Oct 1965
Rubella is clinically
named: RA273 (R=Rubella, A=Abortus, 27=27th fetus, 3=3rd tissue explant)
cultivated on WI-38 cell line.
Thus, at least 47
separate abortions involved in the research for production of present
day rubella vaccine.
(Please note that
the
Roman Catholic Church is of the Devil, because they teach that
sacraments and church membership are required to get to Heaven. The Word
of God says that faith alone in Jesus is all we need to be saved.
Albeit, thankfully, Catholics are some of the strongest pro-life
activists in the world today. Everyone should fight against murderous
abortion.)
Brutal Murder and
dissection of the unborn:
"The Fetus As
Transplant Donor the Scientific, Social, and Ethical Perspectives,"
written by Peter McCullough, who is an immunologist at the University of
Western Australia. It's published by the John Wiley Company in New York.
Here's McCullough. The Australian
International University in Canberra. This is the face page of the book,
and he probably knows more about the subject of using fetal tissue than
anybody in the world. Incidentally, he is pro-life, and it's remarkable
that his book delineates some of the technologies which have formerly
been used to get fetal brain tissue. For example, he talks about how in
Sweden they have been puncturing the sac of a pregnant woman at let us
say 14 to 16 weeks, and then they put a clamp on the head of the baby,
pull the head down into the neck of the womb, drill a hole into the
baby's head, and then put a suction machine into the brain and suck out
the brain cells.
And this is directly from his
book. Healthy human fetuses from 7 to 21 weeks from legal abortions
were used. This is in Sweden. The conceptional age was estimated from
crown rump length and so on. Fetal liver and kidney were rapidly removed
and weighed. Now at 21 weeks, what they were doing, or 18 weeks, or 16
weeks, was what are called prostaglandin abortions. They would inject a
substance into the womb. The woman would then go into mini-labor and
pass this baby. 50% of the time, the baby would be born alive, but that
didn't stop them. They would just simply open up the abdomen of the baby
with no anesthesia, and take out the liver and kidneys.
(Please note that the
Roman Catholic Church is of the Devil, because they teach that
sacraments and church membership are required to get to Heaven. The
Word of God says that faith alone in Jesus is all we need to be
saved. Albeit, thankfully, Catholics are some of the strongest
pro-life activists in the world today. Everyone should fight against
murderous abortion.)
Debi Vinnedge,
Director of Children of God For Life (www.cogforlife.org)
is politically active. Please visit the organization's site to keep up
to date and find out your President's stand on the issue, along with her
correspondence with the manufacturing companies.
|
U.S. Vaccines Derived from Abortion |
|
Diseases |
Vaccine
|
Manufacturer |
Cell line (human fetal) |
|
Polio |
Poliovax |
Aventis-Pasteur |
MRC-5 |
|
Measles, Mumps, Rubella |
MMR II |
Merck & Co. |
RA273 and
WI-38 |
|
Measles-Rubella |
Biavax II |
Merck & Co.
|
RA 273 and
WI-38 |
|
Rubella only |
MR-VAX |
Merck & Co. |
RA 273 and
WI-38 |
|
Rabies |
Imovax |
Vantis-Pasteur |
MRC-5 |
|
Hepatitis A |
Hivrax
Vaqta |
AlaxoSmithKline
Merck & Co. |
MRC-5
MRC-5 |
|
Hepatitis A-B combo |
Twinrix |
GlaxoSmithKline |
MRC-5 |
|
Chickenpox |
Varixax |
Merck & Co. |
WI-38 and
MRC-5 |
|
Smallpox |
Acambix 1000 |
Acambis |
MRC-5 |
|
Ebola |
Unknown (still waiting for trial) |
Crucell & N.I.H. |
PER C6 |
|
HIV |
Unknown (still waiting for trial) |
Merck & Co. |
PER C6 |
|
Sepsis |
Xigris |
Eli Lilley |
HEK 293 |
|
Influenza (flu) |
unknown |
MedImmune |
PER C6 |
About Xigris...
[Published 28 August 2002 Source
Espicom Business Intelligence]
Xigris received approval from the FDA on 21st November 2001 for the
reduction of mortality in adult patients with severe sepsis who have a
high risk of death. Xigris has also been approved in Puerto Rico,
Israel, Australia, Argentina, Peru, Romania, Columbia, Mexico,
Switzerland, India, Singapore and South Africa.
Xigris is a genetically-engineered version of the human activated
protein C molecule, a naturally-occurring protein in the body that helps
to balance many of the forces behind sepsis, including coagulation and
suppression of fibrinolysis. Additionally, patients with severe sepsis
treated with Xigris had a more rapid decline in interleukin-6 levels,
consistent with a reduction in the inflammatory response. The drug is
administered to patients as a 1-time, 96-hour infusion within an ICU
setting. The precise dosage depends on the weight of the patient. In the
PROWESS trial, bleeding was the most common adverse reaction associated
with Xigris therapy. Serious bleeding events, including intracranial
haemorrhage (0.2 per cent in Xigris-treated patients and 0.1 per cent in
placebo-treated patients), were observed during the 28-day study period
in 3.5 per cent of Xigris-treated patients and 2.0 per cent of
placebo-treated patients. The difference in serious bleeding occurred
primarily during infusion.
A human cell line is used in the production of Xigris, as noted under
FDA document, PC 3420 AMP, in the first paragraph, which states:
“Xigris is a recombinant form of
human activated protein C. An established human cell line possessing
the complementary DNA for the inactive human protein C zymogen secretes
the protein into the fermentation medium.”
HEK 293 are cells taken from the kidney of an aborted baby.
About the
293 Cell Line...
The protein C is produced in
the HEK 293 aborted fetal cell line. 293 cells are available from the
American Type Culture Collection. There are variants of the cell
line that derive from the
parent.
ATCC Number:
CRL-1573 Price: $167.00
Designation: 293 Depositors: F.L. Graham
Biosafety Level: 2 Shipped: frozen
Organism: Homo sapiens (human) Morphology: epithelial
Tissue: kidney; transformed with adenovirus 5 DNA
The 293
cell line is a permanent line of primary human embryonal kidney
transformed by sheared human adenovirus type 5 (Ad 5)DNA. [RF32725]
The
cells express the transforming gene of adenovirus 5. Although an earlier
report suggested that the cells contained Adenovirus 5 DNA from both the
right and left ends of the viral genome [RF32764], it is now clear that
only left end sequences are present. [RF50113]
The
cells express an unusual cell surface receptor for vitronectin composed
of the integrin beta-1 subunit and the vitronectin receptor alpha-v
subunit. [RF33793] Purified DNA from this line is available as ATCC
45504 (25 micrograms) and ATCC 45505 (100 micrograms).
The Ad5
insert was cloned and sequenced, and it was determined that a colinear
seqment from nts 1 to 4344 is integrated into chromosome 19
(19q13.2). [RF50113]
United
States Patent 5,681,932
Grinnell October 28, 1997
Inventors: Grinnell; Brian W. (Indianapolis, IN)
Assignee: Eli Lilly and Company (Indianapolis, IN)
Application No.: 458372
Filed: June 2, 1995
 1.
The recombinant human protein C molecule produced by inserting a vector
comprising the DNA encoding human protein C into an
adenovirus-transformed host cell then culturing said host cell under
growth conditions suitable for production of said recombinant human
protein C.
1.
The recombinant human protein C molecule produced by inserting a vector
comprising the DNA encoding human protein C into an
adenovirus-transformed host cell then culturing said host cell under
growth conditions suitable for production of said recombinant human
protein C.
2. The recombinant human
protein C molecule of claim 1 wherein the adenovirus-transformed host
cell is selected from the group consisting of AV12 cells and human
embryonic kidney 293 cells.
3. The recombinant human
protein C molecule of claim 2 wherein the adenovirus-transformed host
cell is an AV12 cell.
4. The recombinant human
protein C molecule of claim 2 wherein the adenovirus transformed host
cell is a human embryonic kidney 293 cell.
About
the 293 Abortion...
In the FDA
report found at
https://www.fda.gov/ohrms/dockets/ac/01/transcripts/3750t1_01.pdf
Key statement...Page 81 lines 14-22
"So the Kidney material, the fetal kidney
material was as follows. The kidney of the fetus was, with an unknown
family history, was obtained in 1972 probably. The precise date is not
known anymore.
The fetus, as far as I can remember was
completely normal. Nothing was wrong. The reasons for the abortion
were unknown to me. I probably knew it at the time, but it got lost,
all this information."
About the PER C6 Abortion and cell
lines....
https://www.fda.gov/ohrms/dockets/ac/01/transcripts/3750t1_01.pdf
Key
excerpts from the above document: Dr. Van Der Eb, Crucel NV is
speaking...
"So
I isolated retina from a fetus, from a healthy fetus as far as could be
seen, of 18 weeks old. There was nothing special with a family history
or the pregnancy was completely normal up to the 18 weeks, and it turned
out to be a socially indicated abortus - abortus provocatus, and that
was simply because the woman wanted to get rid of the fetus."
"The father
was not known not to the hospital anymore, what was written down was
unknown father, and that was, in fact, the reason why the abortion was
requested."
"There was permission, et cetera, and that was, however, was in 1985,
ten years before this. This shows that the cells were isolated in
October 1985, Laeiden University in my lab. At that time already '85, I
should say the cells were frozen, stored in liquid nitrogen, and in 1995
one of these was thawed for the generation of the PER.C6 cells.
"And
this is the final slide just showing you some comparisons between 293
and PER.C6. Again, I remind you that both cell lines were made in my
lab for different reasons. The objective, as I indicated, is for
293--was basic research, and we have done many different transformation
studies after that, not transformation studies, but gene expressions
studies with human embryonic kidney cells in the years following that up
to now, I would say.
"PER
C6 was made JUST FOR PHARMACEUTICAL MANUFACTURING OF ADENOVIRUS VECTORS
As to RCA free, PER.C6 are RCA Free. The history documentation of the
cell line has been carried out completely for PER. C6 and was not done
at that time for 293.
"And then pharmaceutical industry
standards... I realize that this sounds a bit commercial, but PER C6
were made for that particular purpose."
If you find this document no
longer available, click
here
About the Cell Lines and Adenovirus
(AD5) Safety issues...
https://www.fda.gov/ohrms/dockets/ac/01/briefing/3750b1_01.htm
Background
In
1954, during discussions surrounding the development of adenovirus
vaccines for use in the military, the U.S. Armed Forces Epidemiology
Board (AFEB) recommended the use of "normal cells" as the substrate for
vaccine production rather than cell lines established from human tumors.
This decision was based on concerns about the possibility that human
tumor cells might be contaminated with occult oncogenic agents that
might be transferred to vaccine recipients, an event which might in turn
increase the risk of cancer and other neoplastic diseases in vaccinees.
As evidenced by current regulatory guidelines and activities of control
authorities worldwide, the precedent set in 1954 by the AFEB remains an
important factor in the acceptance of all substrates for vaccine
manufacture. Currently, the only cultured animal cells that have been
used as substrates in U.S. licensed viral vaccines have been primary
cells (e.g., derived from monkey, chick, mouse), diploid cell lines
(e.g., WI38, MRC-5, FRhL-2), or immortalized (continuous), non-tumorigenic
cell lines (e.g., VERO).
Over
the past 47 years, two important factors have emerged that warrant
serious consideration of the use of immortalized tumorigenic cell lines
for viral vaccine production. The first of these factors is that certain
novel virus vectors that are presently under development for
high-priority target diseases, most notably AIDS, cannot feasibly be
propagated in
traditionally acceptable cell substrates. The second factor is that
scientific understanding of neoplastic processes and viral-induced
carcinogenesis has rapidly advanced, as has the ability to detect and
identify infectious, oncogenic agents and other types of adventitious
agents that may potentially contaminate cell substrates. These factors
underscore the need for developing a regulatory framework in which the
relative benefits and risks in using tumorigenic cell lines for vaccine
production can be carefully and cautiously revisited.
FDA
would like the VRBPAC to consider the potential risks in using two novel
cell substrates, 293 cells and PER.C6 cells. These cell lines were
developed by transforming human embryonic kidney cells (293) and human
embryonic retinal cells (PER.C6) with the transforming early region 1
(E1) of adenovirus type 5 (Ad5). Since cell lines such as 293 and PER.C6
express the Ad5 E1 region, they are able to complement the growth of
defective Ad5 vectors which have been "crippled" by deletion of E1.
Defective Ad5 vectors have been engineered to express foreign genes such
as those from human immunodeficiency virus (HIV), the causative agent of
AIDS, and vectors of this type are thought to have significant potential
for vaccine development because of their demonstrated ability to
generate cell-mediated immune responses to HIV. However, a feature of
regulatory importance associated with Ad5-transformed cells is their
capacity to form tumors in immunodeficient animals such as nude mice.
In
considering potential risks associated with the use of these so-called
Designer Cell Substrates – i.e., neoplastic cells derived from
normal human cells transformed by defined viral or cellular oncogenes or
by immortalizing cellular genes (e.g., telomerase) – OVRR/CBER is
considering the approach outlined below within the framework of a
"defined-risks" assessment (see enclosed reference Lewis et al.,
"A defined-risks approach to the regulatory assessment of the use of
neoplastic cells as substrates for viral vaccine manufacture", In
Evolving Scientific and Regulatory Perspectives on Cell Substrates for
Vaccine Development. Brown, Lewis, Peden, Krause (eds.) Develop. Biol.
Stand. [in press]). This framework is intended to examine, and wherever
possible, to quantify the potential risk of "transmitting" the
tumorigenic components of the cell substrate used for vaccine
production, and determine whether that "transmission" might pose a risk,
particularly an oncogenic risk, to vaccinees. Factors that could
influence the risk associated with the use of Designer Cell Substrates
include (1) the known mechanism of cell transformation leading to the
development of tumorigenic cells; (2) residual cell substrate DNA; and
(3) the presence of adventitious agents, especially oncogenic viruses.
These three factors are discussed in more detail below.
Tumorigenicity of Adenovirus 5-Transformed Designer Cell Substrates
The
purpose of tumorigenicity testing as applied to cell substrates used for
viral vaccine manufacture is to discriminate between cells that have the
capacity to form tumors and cells that do not form tumors. The potential
risk of oncogenic activity is thought to be higher for cell substrates
that have the capacity to form tumors, whereas the potential risk is
thought to be low for cell substrates that are unable to form tumors. In
considering the risk of tumorigenicity of Ad5-transformed Designer Cell
Substrates, it is important to consider the molecular processes that
determine the ability of the cells to form tumors.
Adenovirus 5 does not produce tumors when injected into rodents, but it
does transform rodent cells in tissue culture. Like adenovirus 5 virions,
adenovirus 5-transformed cells do not produce tumors when injected into
immunocompetent adult rodents, but these cells can form tumors when
injected into immunodeficient rodents such as nude mice. The
tumor-forming capacity of Designer Cell Substrates that are produced by
transforming normal human cells with adenovirus 5 can be evaluated by
comparing them with adenovirus 5-transformed rodent cells. The
adenovirus 5 early region 1 (E1) is composed of the transcription units
E1A and E1B, which transform normal cells to neoplastic cells through a
multi-step process. The E1A transcription unit immortalizes the cells
and establishes those characteristics of the transformed cells that
permit them to be eliminated by the antitumor defenses of
immunocompetent rodents. During the transformation process, E1A
sensitizes cells to apoptosis (programmed cell death) and increases
their susceptibility to killing by natural killer cells, macrophages,
and cytotoxic lymphocytes, as well as cytokines such as tumor necrosis
factor (see Routes et al., 2000a, 2000b). The adenovirus E1B
region alone is unable to immortalize cells, but its function during
neoplastic transformation ensures cell survival by inhibiting
virus-induced cell killing. Thus, the E1A region immortalizes cells and
determines their limited capacity to form tumors in immunodeficient
rodents, whose antitumor immune defenses are compromised. The complexity
of these tumor-host processes and their action through nontransferable,
immune mechanisms of the host implies that the capacity of adenovirus 5
E1-transformed mammalian cells to form tumors in immunodeficient rodents
does not represent a risk factor for the manufacture of viral vaccines
provided the cells can be shown to be devoid of adventitious agents (see
additional discussion on adventitious agents below).
Several approaches can be considered in evaluating tumorigenicity of
adenovirus 5-transformed human cell substrates. These approaches include
demonstration that the tumor-forming capacity of the cells in rodents is
adenovirus 5-like, and that the cells in the master cell bank are devoid
of known and occult adventitious agents.
Potential Risks of DNA in Vaccines
Residual DNA in vaccines derived from tumorigenic cells, including those
transformed by Ad5, can pose potential risks to the vaccine recipient in
two respects: oncogenicity and infectivity. Each of these biological
properties must be considered and evaluated for each cell substrate.
The
oncogenic risk of cell substrate DNA has been considered to be due to
several mechanisms. First, the residual DNA could have dominant
activated oncogenes that could exert their effect following expression
in recipient cells. In the case of Ad5-transformed cells, the dominant
oncogenes would include the E1A and E1B genes. Second, the incoming DNA
could integrate into the host genome in certain genes, such as the p53
gene or the retinoblastoma susceptibility (RB) gene, termed tumor
suppressor genes, which are involved in cell cycle control among other
cellular processes. Loss of function of tumor suppressor genes has been
associated with certain human tumors. Third, integration of residual
cell-substrate DNA could result in the activation of cellular regulatory
genes by promoter/enhancer insertion, and this could result in the
development of a neoplastic phenotype; this mechanism for tumor
development was initially described in chickens for leukemia formation
by avian leukosis viruses. Another result of integration that has been
described is an increased methylation of adjacent DNA sequences as well
as sequences on other chromosomes, although the consequences of such
changes in methylation patterns to a cell are unknown.
The
second biological activity of DNA that should be considered is its
potential infectivity. If a genome of a DNA virus or the provirus of a
retrovirus is present in the cell substrate used for vaccine
manufacture, then the residual DNA has the potential, upon inoculation
into the vaccine recipient, to produce infectious virus from this DNA
and thus establish a productive infection.
The
assessment of the risk of DNA — both the oncogenic risk and the
infectious risk — needs to be considered both in terms of (1) the amount
of residual DNA inoculated; and (2) the concentration of oncogene or
infectious genome present in this DNA. One assumption is that the
biological activity of any DNA administered is directly proportional to
the amount of that DNA, and if the active component (oncogene or
infectious genome) is carried as part of the cell-substrate DNA, the
amounts of the oncogene or infectious genome will be present at a level
of 10-5 to 10-6. This is because the haploid
mammalian genome is 3 x 109 base pairs, whereas an average
gene is between 3 x 103 and 104 base pairs. Thus,
if the residual DNA is present at 10 ng, an oncogene in that DNA would
be present at between 0.00001 and 0.0001 ng. Currently there are no data
indicating that purified isolated oncogenes or any other DNA are
biologically active at these levels.
It
is also important to note that an additional safety margin for oncogenic
activity is provided by the multi-step nature of cancer. This is because
if more than one gene or event is required, the risk is diminished and
is given by the product of the risk for each event. Thus, if the risk of
a neoplastic event being induced by one oncogene is 1 in 106,
then if two oncogenes are required, the risk is reduced to 1 in 1012.
Strategies that can be considered in evaluating residual DNA for vaccine
products manufactured in adenovirus 5-transformed cell substrates
include restricting the level of residual DNA to 10 ng or less. If these
levels of residual DNA are not feasible, other methods can be considered
to demonstrate the safety of higher levels of DNA, such as the
inoculation of cell-substrate DNA into neonatal rodents. In the future,
more sensitive animal models and in vitro assays may be developed to
assess the oncogenic activity of DNA.
The
experience in the early 1960s with SV40 contamination of poliovirus and
adenovirus vaccines and the continuing questions regarding whether SV40
could be responsible for some human neoplasms underscore the importance
of keeping viral vaccines free of adventitious agents. This is
particularly important when there is a theoretical potential for
contamination of a vaccine with viruses that might be associated with
neoplasia.
It
is unclear whether neoplastic cells have a greater or lower adventitious
agent risk than other types of cells. Because they can be grown for long
periods in tissue culture, there may be greater opportunities for any
adventitious agents to be detected. Because neoplastic cells survive
indefinitely, it is easier to qualify and bank cells that have passed
all tests, especially as compared with primary cells (which are derived
repeatedly from live tissue and must be re-qualified with each use).
Moreover, many neoplastic cells can be grown in serum-free medium,
potentially reducing the likelihood of contamination with bovine
adventitious agents. However, if their growth in tissue culture is not
well controlled, there may exist additional opportunities for
contamination of cells with a longer lifespan. In cases of neoplastic
cells for which the transforming event is unknown, there is also a
theoretical possibility that transformation occurred as a result of a
previous viral infection. Because some mammalian tumors and some cells
transformed by viruses contain infectious virus, cells transformed by an
unknown mechanism have a theoretical risk of containing a transforming
virus. Cells for which specific knowledge of the transforming event
exists (and can be shown not to be a virus that persists in the cells)
may be more easily characterized than cells for which there is no
specific knowledge of the transforming event (which could theoretically
have been due to an infection with a known or an unknown virus).
Extensive adventitious agent testing is required for all cells that are
proposed for use in vaccine production. This includes testing in various
tissue culture systems, inoculation of animals followed by observation
or detection of pathogen-specific antibodies, observation by electron
microscopy, and molecular tests as is appropriate based on the history
and type of cell to be tested. Specific polymerase chain reaction assays
are used to rule out the presence of many different viruses. PCR-based
reverse transcriptase assays are used to rule out the presence of
retroviruses. The most sensitive of these assays include amplification
steps, either as a result of viral growth in culture or in an organism,
or molecular amplification such as in PCR.
Although Ad5-transformed cells are thought to be transformed by a known
mechanism, the consequences of overlooking an occult oncogenic agent are
significant. As these are the first cells in their class to be
considered for vaccine production, evaluating them for the presence of
occult oncogenic agents could enhance confidence in their use. One
relevant animal assay that could be used for such an evaluation is the
inoculation of cell-free lysates into susceptible newborn rodents,
followed by observation for 5-6 months. This assay would detect most
known tumor viruses, as well as potentially detect unknown tumor
viruses.
Several promising areas of research suggest that experimental assays to
detect unknown adventitious agents could soon become more generally
available. As such assays become available, they could be considered for
use in qualifying novel cell substrates, including neoplastic cell
substrates.
Risk of Transmissible Spongiform Encephalopathy (TSE)
In
addition to assessing the possibility of contamination of cell
substrates with infectious virus, it is important to consider other
agents such as the agent of TSE. There are several mechanisms by which
vaccine cell substrates, including neoplastic cells, could theoretically
become infected with a TSE agent. First, viral vaccines are developed
and manufactured in cell substrates that may be derived from humans, and
all human cells represent a finite possibility of being derived from
individuals with a propensity to develop sporadic or familial
Creutzfeld-Jakob disease (CJD). Although the mechanism by which such
individuals develop CJD is not understood, CJD has in the past been
transmitted to humans by biological products derived from CJD patients,
such as human growth hormone and dura mater grafts. Second, vaccine cell
substrates are usually exposed to products derived from cattle during
tissue culture. Bovine spongiform encephalopathy (BSE) has been
transmitted to humans in Europe in the form of variant CJD, possibly due
to ingestion of infected beef. Under certain circumstances, cells in
tissue culture can support the replication of certain TSE agents,
although it is not known whether human cells in tissue culture
can sustain the replication of the BSE agent. Assays to detect
TSE/BSE agent contamination exist, but they may not be sensitive enough
to exclude low levels of contamination. Until more is known about the
replication of TSE/BSE agents in tissue culture and until more sensitive
assays to detect these agents become available,
the concern over the
possible contamination of viral vaccines with TSE/BSE agents will be at
least a theoretical
consideration in vaccine development.
The
use of immortalized, neoplastic human cells as substrates to develop
recombinant viral vectors as vaccines also raises theoretical concerns
with regard to possible contamination with TSE/BSE agents. These
concerns include: (1) the implications of the possible presence of a
prion protein (PrP)-encoding gene (PRNP) that is abnormal in the
individual from whom the cells were derived; (2) the possibility that
the genomic instability attendant with neoplastic processes could
produce pathogenic alterations in the normal PRNP gene; (3) the possible
exposure to agents of BSE present in bovine serum used in the
propagation of the cells; (4) the possibility that an increased level of
expression of either a normal or abnormal PRNP gene or other unknown
factors in neoplastic cells might, upon exposure, sustain the
replication of abnormal PrP proteins or otherwise contribute to the
development of TSE in humans; and (5) the possibility that differences
in the levels of expression of PRNP genes among clonal/subclonal
populations of neoplastic cells may make evaluation of these potential
risks more difficult. Since the lifetime risk of sporadic TSE in the
population is about one case per ten thousand people (see Brown, P. et
al., [1985], Potential epidemic of Creutzfeldt-Jakob disease from human
growth hormone therapy. N Engl J Med. 1985, 313:728-731), there is a
finite risk that random tissue samples used for the development of
neoplastic cell lines could contain abnormalities that might be
associated with TSE transmission.
Several strategies can be considered for evaluation of neoplastic human
cells for possible contamination with TSE/BSE agents. These strategies
include a determination of the origin of the cells with respect to the
possible family history and medical history of the donor regarding TSE
risk factors and the identification and documentation of possible
exposure of the cell line to bovine-derived materials, such as fetal
bovine serum from countries with BSE. Further, two validated methods
that could be used to evaluate the potential risk from the TSE agent
include sequencing of the PRNP gene from neoplastic cell substrates and
evaluating all such cell substrates by Western blot for the presence of
protease resistant PrP. Additional studies may become feasible in the
near future and may include evaluation of PRNP expression levels,
determination of the ability of a cell substrate to support replication
of the BSE agent, and evaluation of for the presence of infectious TSE
agents by animal inoculation. As new assays for the detection and
evaluation of TSE agents become available, they should be introduced as
appropriate for cell substrate screening.
OVRR/CBER
plans to present these issues to the FDA TSE Advisory Committee for a
comprehensive discussion in the near future. In the interim, for cell
substrates for which the presence of TSE could be a risk, sponsors
should evaluate this issue by a combination of strategies, as may be
technically feasible. Nevertheless, OVRR/CBER would like this Committee
to be aware of and consider those issues related to the possible
presence or exposure of cell substrates used for the development of
viral vaccines to agents associated with TSE/BSE.
SUMMARY
Recent animal experiments have demonstrated the utility of Ad5 vaccine
vectors as means of stimulating cell-mediated immunity against HIV-1.
Based on the review of available data, OVRR/CBER believes that there is
an extremely low probability that residual DNA from the adenovirus
5-transformed human cells could transfer traits that could induce
neoplastic development in vaccinees. OVRR/CBER also believes that such
cells may be considered for the development of HIV vaccines, provided
that the phenotype of the cells can be documented to be of an adenovirus
5 E1 type, and that appropriate testing rules out the presence of
adventitious agents within the limits of state-of-the-art technology.
 The Chickenpox,
Hepatitis-A and MMR vaccines were developed using aborted fetal cell
lines, MRC-5 and WI-38. This has never been hidden from the
public. When parents take their children to the doctor for inoculations,
who asks for the package inserts? Who asks for a list of the vaccine
ingredients?
Most parents
want to know the risks and possible side effects. Parents are mostly
concerned with the health of their children as some doctors are.
Doctors have been administering the vaccines for years. Yet, how many
of them have ever checked into the ingredients? This has always been
right at their fingertips. What would happen if they did? They would
read that the vaccine contains "MRC-5", "WI-38" (or both) "human diploid
cell lines".
The Chickenpox,
Hepatitis-A and MMR vaccines were developed using aborted fetal cell
lines, MRC-5 and WI-38. This has never been hidden from the
public. When parents take their children to the doctor for inoculations,
who asks for the package inserts? Who asks for a list of the vaccine
ingredients?
Most parents
want to know the risks and possible side effects. Parents are mostly
concerned with the health of their children as some doctors are.
Doctors have been administering the vaccines for years. Yet, how many
of them have ever checked into the ingredients? This has always been
right at their fingertips. What would happen if they did? They would
read that the vaccine contains "MRC-5", "WI-38" (or both) "human diploid
cell lines".  One
if the most highly misunderstood notions among moral theologians and
ethicists is that the abortions involved were not done with the
intention of creating vaccines. In fact, in response to President
Bush’s decision on federal funding for embryonic stem cell research (ESCR),
the USCCB highlights this point as follows:
One
if the most highly misunderstood notions among moral theologians and
ethicists is that the abortions involved were not done with the
intention of creating vaccines. In fact, in response to President
Bush’s decision on federal funding for embryonic stem cell research (ESCR),
the USCCB highlights this point as follows:




